Written by Khalid al Zubi M.D and Khalil alsalem M.D
Glaucoma is a group of eye diseases associated with acute or chronic destruction of the optic nerve with or without concomitant increased intraocular pressure (IOP). In the US, glaucoma is the second leading cause of blindness in adults following age-related macular degeneration (AMD). The two main types are open-angle glaucoma and angle-closure glaucoma. Open-angle glaucoma accounts for 90% of all cases of glaucoma, is slowly progressive, and is initially often asymptomatic, but leads to bilateral peripheral vision loss over time.
With appropriate treatment that lowers IOP (e.g., topical prostaglandins), progression can be stopped before severe damage occurs. Acute angle-closure glaucoma, on the other hand, is characterized by the sudden onset of a painful, red, and hard eye in combination with frontal headache, blurry vision, and halos appearing around lights. Immediate initiation of medical therapy (e.g., timolol eye drops and IV acetazolamide) is crucial to decrease IOP and prevent vision loss rapidly. Chronic angle-closure glaucoma manifests and is managed similarly to open-angle glaucoma.
Glaucoma Epidemiology
- Second leading cause of blindness in adults in the US following age-related macular degeneration (AMD)
- ∼ 2.3 million cases of glaucoma in the US
- Vision impairment in ∼ 10% of patients
- Blindness in ∼ 5% of patients
- Open-angle glaucoma is more common than angle-closure glaucoma
| types of glaucoma | Open-angle glaucoma | Angle-closure glaucoma |
| Risk factors | Age > 40 years, African descent Diabetes mellitus Myopia Family history of glaucoma | Asian descent Mydriasis (like atropine) |
| Clinical features | often asymptomatic Bilateral progressive visual field loss | Sudden onset Unilateral red, hard, and severely painful eye Frontal headaches Vomiting, nausea blurred vision Dilated, nonreactive pupil |
| Treatment | Prostaglandin eye drops laser trabeculoplasty surgical trabeculectomy | Topical timolol apraclonidine and IV acetazolamide Curative treatment is laser peripheral iridotomy No mydriatic drugs! |
| Prognosis | good if early diagnosed blinding occurs many years after if left untreated | very good if treated early, can lead to central retinal artery or central retinal vein occlusion of left untreated |
| Decrease synthesis of aqueous humor | |
| Beta blocker, like Timolol , Carteolol | Blockage of sympathetic nerve fibers in the ciliary epithelium |
| Alpha 1 agonist like Epinephrine | Via decrease in cAMP Additional conjunctival vasoconstriction |
| Alpha 2 agonists like Apraclonidine, Brimonidine | Via decrease in cAMP |
| Carbonic anhydrase inhibitors like Acetazolamide (oral)Methazolamide (oral)BrinzolamideDorzolamide | Inhibits the action of carbonic anhydrase enzyme |
| Increase Aqueous humor outflow | |
| Prostaglandin analogues like Latanoprost, Travaprost, Bimatoprost | Decreases resistance through uveoscleral flow |
Glaucoma Pathophysiology
- Aqueous humor is produced by the ciliary body on the iris, flows from the posterior chamber through the pupil into the anterior chamber, and then drains back into the venous system via the trabecular meshwork in the angle of the anterior chamber.
- Physiologically, the flow of aqueous humor against resistance generates an average intraocular pressure (IOP) between 10–21 mm Hg.
- Any process that disrupts the flow of aqueous humor (e.g., adhesion of the iris to the lens) may increase intraocular pressure and lead to optic nerve damage and visual impairment.
Open-angle glaucoma
Definition
- Open-angle glaucoma (also chronic glaucoma): generally bilateral, progressive loss of optic nerve fibers with open chamber angles (often with increased intraocular pressure), not caused by another systemic or local condition
Etiology
- Primary cause unclear
- Risk factors
- Age > 40 years
- Increased IOP
- European or African descent
- Diabetes mellitus
- Familial predisposition
- Myopia
- Steroid use
Pathophysiology
- Secondary clogging of the trabecular meshwork or reduced drainage → gradual increased intraocular pressure → vascular compression → ischemia to the optic nerve → progressive visual impairment.
- Causes
- Secondary clogging due to
- Inflammatory cells (e.g., uveitis)
- Red blood cells (e.g. vitreous hemorrhage)
- Material from retinal detachment in the aqueous humor
- Reduced drainage due to
- Increased episcleral venous pressure
- Damaged trabecular meshwork after a chemical injury
- Secondary clogging due to

Clinical features
- Initially often asymptomatic
- Over time, nonspecific symptoms such as mild headaches, impaired adaptation to darkness
- Generally bilateral, progressive visual field loss (from peripheral to central)
- Arcuate scotoma: arch-shaped scotoma that starts from the blind spot
Diagnostics
- Slit-lamp examination of the anterior segment: normal appearing anterior chamber angle
- Tonometry: to measure increased intraocular pressure (standard values range between 10–21 mm Hg)
- increased intraocular pressure may be seen in up to 40% of patients with primary open-angle glaucoma but is not a requirement for diagnosis
- Gonioscopy: to rule out angle-closure glaucoma
- Fundoscopy: cupping and pallor of optic disc, disc hemorrhage, diffuse or focal narrowing of the optic disc rim
Treatment of open-angle glaucoma
- Indicated in all patients diagnosed with open-angle glaucoma (even if asymptomatic)
- Options include medical therapy, laser surgery, and open surgery
- Topical prostaglandins are most effective and usually used initially; other drugs (with a different mechanism) may be added if topical prostaglandins are unsuccessful.
- No decrease in IOP with one drug: Discontinue and replace with another drug or treatment option.
- Partial response to one drug: Consider combination therapy with other glaucoma medications or switch to an alternative single-agent therapy.
- Goal of therapy (target IOP): ≥ 25% decrease in pretreatment IOP
Pharmacotherapy
The following regimen is the most commonly followed and is also effective in patients with chronic angle-closure glaucoma refractory to laser peripheral iridotomy.
- Important considerations
- Tailor regimen to the patient’s comorbidities and tolerance.
- Adherence to pharmacotherapy is crucial.
- Advise patients to occlude their nasolacrimal ducts following the topical administration of drugs.
- Patients should be continually assessed for disease progression and for local and systemic side effects.
- Preferred first-line therapy: topical prostaglandin analogs
- Latanoprost
- Travoprost
- Bimatoprost
- Alternative options
- Topical beta blockers alone and/or alpha-2 agonists
- Beta blockers
- Timolol
- Betaxolol
- Alpha-2 agonists
- Brimonidine
- Apraclonidine : generally used perioperatively in patients with refractory glaucoma [5]
- Beta blockers
- Topical carbonic anhydrase inhibitors
- Brinzolamide
- Dorzolamide
- Topical beta blockers alone and/or alpha-2 agonists
- Refractory glaucoma: oral carbonic anhydrase inhibitors
- Acetazolamide
- Methazolamide
Interventional therapy
Procedures that lower IOP by facilitating drainage of aqueous humor
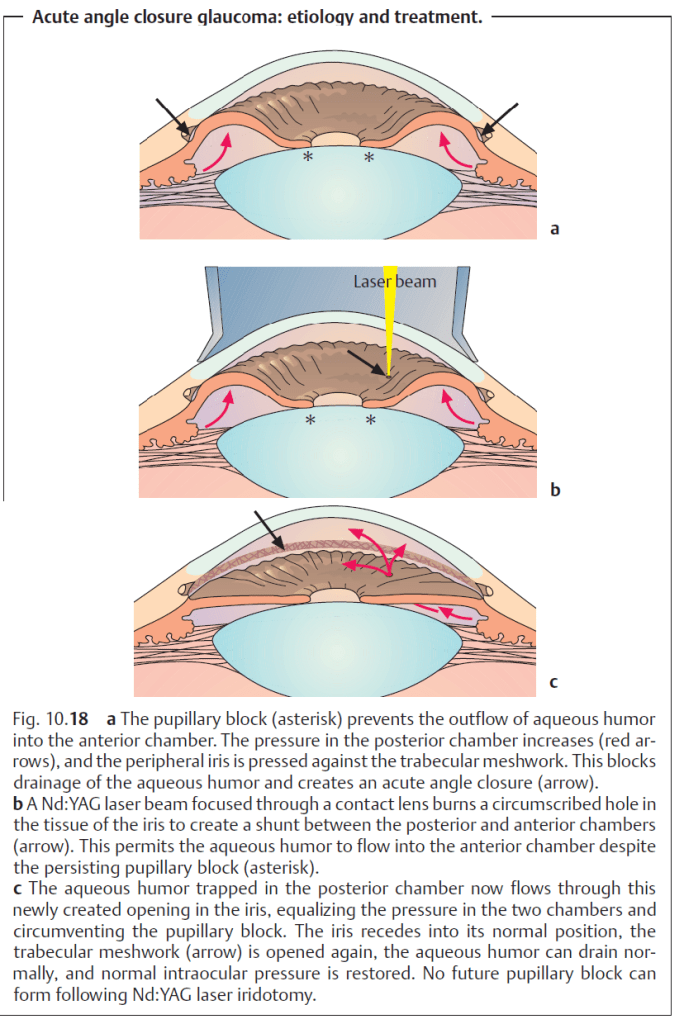
- Laser trabeculoplasty
- Indications
- An alternative first-line treatment for patients with advanced disease at presentation
- Glaucoma refractory to pharmacotherapy
- Patients nonadherent or intolerant to pharmacotherapy
- Procedure: use of a laser to thermally ablate the trabecular meshwork cells and improve aqueous outflow
- Alternative: selective laser trabeculoplasty
- Pigmented cells of the trabecular meshwork are selectively targeted for thermal ablation.
- Has a better safety profile than laser trabeculoplasty and may soon become the preferred first-line therapy for treatment of open-angle glaucoma
- Indications
- Surgical trabeculectomy
- Indications: the same as those for laser trabeculoplasty
- Procedure: involves the creation of a tunnel (through excision of trabecular meshwork) from the anterior chamber to the subconjunctival space under a thin scleral flap
- Tube shunt surgery
- Indication: glaucoma refractory to trabeculectomy
- Procedure: A small silicone tube is inserted into the anterior chamber of the eye through which aqueous humor is drained into a valved chamber that is placed on the sclera underneath the upper eyelid.
Procedures that lower IOP by decreasing aqueous humor production
- Cyclodestructive surgery
- Indication: glaucoma refractory to other treatment options
- Procedure: laser or cryosurgical destruction of the ciliary body
Prevention
- General screening for glaucoma is not considered cost-effective but is currently recommended in the following patient groups:
- Personal history of diabetes mellitus
- Family history of glaucoma
- African Americans > 50 years of age
- Hispanic Americans > 65 years of age



Angle-closure glaucoma
Definition
- Angle-closure glaucoma (also closed-angle glaucoma): sudden and sharp increase in intraocular pressure caused by an obstruction of aqueous outflow (most commonly as a result of an occlusion of the iridocorneal angle ) and associated with optic neuropathy and visual field defects
- Acute angle-closure glaucoma (AACG): sudden obstruction of the iridocorneal angle causing a rapid, acutely symptomatic, and vision-threatening increased intraocular pressure, often > 30 mm Hg
- Chronic angle-closure glaucoma: chronic obstruction of the iridocorneal angle with peripheral anterior synechiae resulting in an insidious and progressive increased intraocular pressure that typically remains asymptomatic until glaucomatous optic neuropathy and irreversible visual field defects have developed
- Angle-closure suspect: normal IOP with iridotrabecular contact is seen on gonioscopy
Etiology/risk factors
- Anatomic features predisposing to angle closure: shallow anterior chamber (e.g., hyperopia, short eye)
- Advanced age
- Female gender
- Asian or Inuit ethnicity
- Eye injury with scarring and adhesions
- Rubeosis iridis
- Mydriasis
- Drug-induced: anticholinergics (e.g., atropine) , sympathomimetics, decongestants
- Darkness
- Stress/fear response

Pathophysiology
- Common pathophysiology of angle-closure glaucoma: blockage of the trabecular meshwork → decrease drainage of aqueous humor from the eye → increased intraocular pressure
- Primary angle-closure glaucoma: glaucoma due to an anatomical variant of ocular structure(s) that narrows the iridocorneal angle and increases the likelihood of trabecular meshwork obstruction
- Shallow chamber depth (enhanced by mydriasis)
- Small anterior segment of the eye
- Plateau iris configuration
- Most common cause of angle-closure glaucoma in individuals < 50 years of age [19]
- An anatomical variant in which the iris is abnormally inserted in a more anterior position onto the ciliary body and lies on a horizontal plane (compared to its normal slightly convex plane), thus crowding the trabecular meshwork.
- The anterior chamber depth remains normal.
- Secondary angle-closure glaucoma: glaucoma due to acquired conditions that occlude the iridocorneal angle with/without a pupillary block
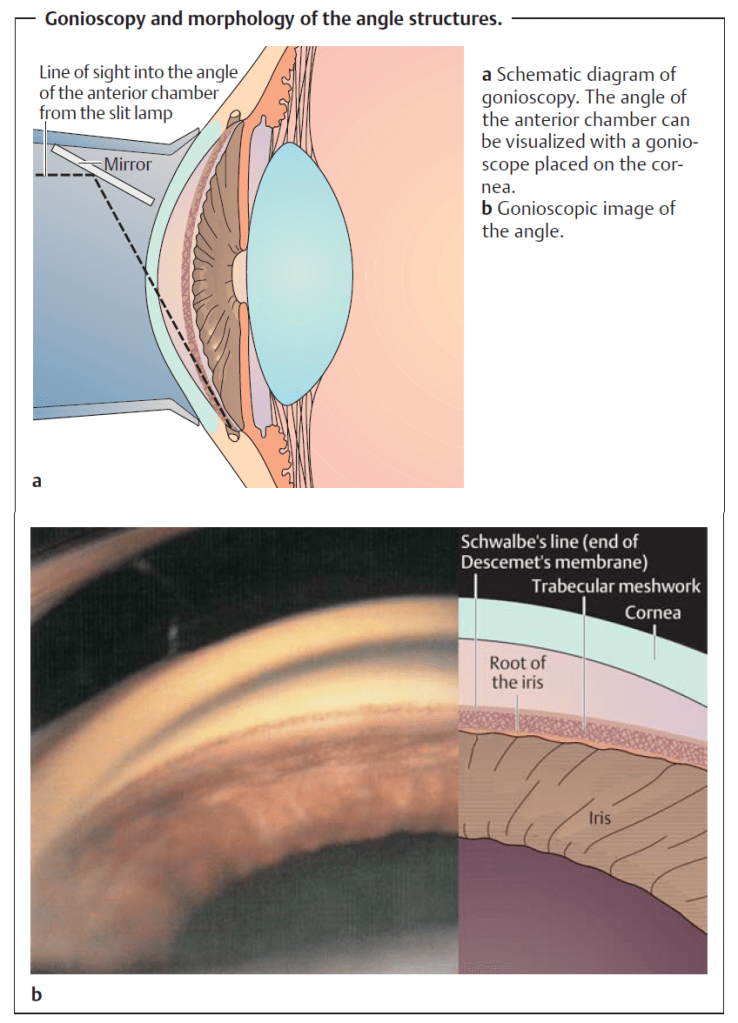
- Pupillary block → increased intraocular pressures in the posterior chamber of the eye → iris bulging forward → peripheral iris pressing against the cornea → narrowing of iridocorneal angle (anterior chamber angle) → blockage of the trabecular meshwork
- Examples of conditions causing pupillary block
- Inflammatory conditions (e.g., uveitis) causing posterior synechiae between the iris and lens
- Anterior dislocation of the lens (ectopia lentis)
- Enlargement of the lens (e.g., mature cataract)
- Examples of conditions causing pupillary block
- Direct blockage of the trabecular meshwork (i.e., without a pupillary block). Examples include:
- Inflammatory conditions causing peripheral anterior synechiae between the iris and cornea
- Rubeosis iridis (neovascular glaucoma): hypoxia and the release of vasoproliferative substances (common in retinal ischemia due to central retinal vein occlusion or in diabetes mellitus) → angiogenesis of the iris and the ciliary body → narrowing of the anterior chamber angle
- Pupillary block → increased intraocular pressures in the posterior chamber of the eye → iris bulging forward → peripheral iris pressing against the cornea → narrowing of iridocorneal angle (anterior chamber angle) → blockage of the trabecular meshwork
Clinical features
- Acute angle-closure glaucoma
- Sudden onset of symptoms
- Unilaterally inflamed, reddened, and severely painful eye (hard on palpation)
- Frontal headaches, vomiting, nausea
- Blurred vision and halos seen around light
- Cloudy cornea (opacification)
- Mid-dilated, irregular, unresponsive pupil
- Complications: rapid permanent vision loss due to ischemia and atrophy of the optic nerve
- Chronic angle-closure glaucoma
- Asymptomatic in early stages
- Progressive vision loss beginning with peripheral fields of vision (due to gradually increasing optic nerve compression)
Acute angle-closure glaucoma is a medical emergency, as it can cause permanent vision loss if left untreated!

Diagnostic methods for glaucoma
Approach
Acute angle-closure glaucoma is vision-threatening and requires emergency ophthalmology evaluation as soon as the clinical diagnosis is suspected.
- Both eyes should be evaluated even if symptoms are unilateral.
- A clinical diagnosis of angle-closure glaucoma is confirmed with the following findings:
- Increased intraocular pressure (> 21 mm Hg): on tonometry
- Narrowing/closure of the iridocorneal angle (i.e., iridotrabecular contact): on gonioscopy or slit -lamp examination
- Tests to assess for glaucomatous damage should be performed in all patients.
- Optic disc changes (slit-lamp examination or undilated fundoscopy)
- Visual acuity
- Visual field testing
- Provocative testing (e.g., placing the patient in a dark room, administering mydriatics) is not recommended in acute angle-closure glaucoma because it is time-consuming, exacerbates symptoms, and is of questionable clinical significance.
- Other causes of painful red eye (especially uveitis) and/or headache with ocular pain (e.g., migraine) should be considered if diagnostic findings are inconclusive.
Do not use mydriatic drugs (e.g., atropine and epinephrine) during ophthalmologic examination in patients with acute angle-closure glaucoma! Moreover, do not cover the eye, since darkness induces mydriasis and worsens the condition!
Tonometry
- Indication: all patients with suspected glaucoma
- Procedure: measurement of IOP by placing a probe over the cornea .
- Characteristic findings [11][13]
- Acute angle-closure glaucoma: IOP is typically > 30 mm Hg
- Chronic angle-closure glaucoma: IOP > 21 mm Hg
- Angle-closure suspect: normal IOP
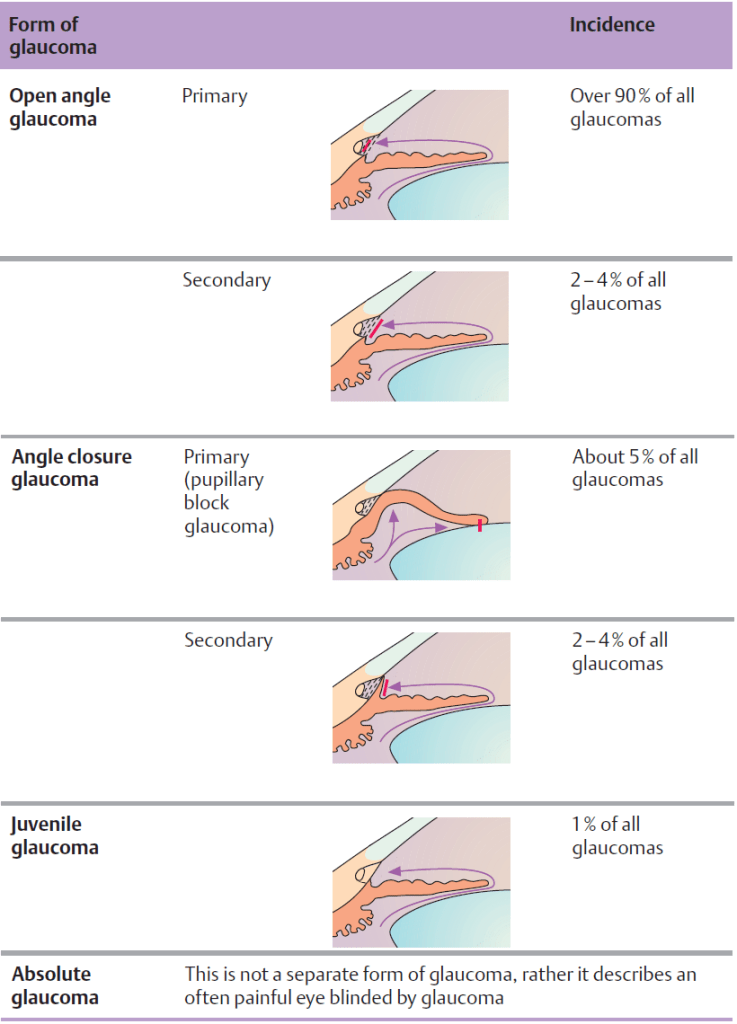
Gonioscopy
- Indications
- Gold-standard test to assess the iridocorneal angle in suspected angle-closure glaucoma
- To distinguish between primary and secondary causes of angle closure (see ”Pathophysiology”)
- Procedure: A special lens is placed directly on the eye and the iridocorneal angle is visualized directly using a slit lamp.
- Typically conducted by a trained specialist
- Should be performed on both eyes
- Characteristic findings
- Narrowing or closure of the iridocorneal angle (i.e., ≥ 180º iridotrabecular contact)
- Etiology of narrowed/closed iridocorneal angle may be apparent, such as:
- Peripheral anterior synechiae
- Plateau iris configuration
Slit-lamp examination
- Indication: to evaluate the anterior chamber and optic disc in all patients with suspected glaucoma
- Supportive findings
- Acute and chronic angle-closure glaucoma: shallow anterior chamber
- Acute angle-closure glaucoma
- Cornea: cloudy or hazy
- Pupil: mid-dilated (4–6 mm); sluggish pupillary reaction
- Chronic angle-closure glaucoma: signs of glaucomatous optic neuropathy
- Increased cup-to-disc ratio > 0.5
- Asymmetrical cup-to-disc ratio between eyes
- Superficial hemorrhages within the optic disc
- Focal thinning and pallor of the neuroretinal rim
Direct fundoscopy (with undilated pupils)
- Indication: an alternative to slit-lamp examination to assess for optic disc damage
- Supportive findings
- Acute angle-closure glaucoma: edema and microhemorrhages of the optic disc with or without signs of glaucomatous optic neuropathy
- Chronic angle-closure glaucoma: signs of glaucomatous optic neuropathy
Do not dilate the pupils to evaluate the fundus in suspected glaucoma!
Visual acuity
- Indication: all patients with glaucoma
- Supportive findings
- Acute angle-closure glaucoma: Corneal edema may decrease visual acuity even in the absence of glaucomatous optic neuropathy.
- Chronic angle-closure glaucoma: There may be decreased central vision or complete blindness in advanced disease.
Visual field testing
- Indication: all patients with glaucoma
- Techniques
- Confrontation visual field exam: preferred in the ER when an ophthalmologist is not immediately available
- Automatic static perimetry: preferred if an ophthalmologist is available
- Characteristic findings
- Glaucomatous visual field defects: a characteristic pattern of visual field defects as a result of glaucomatous optic neuropathy
- Early-stage: arcuate or double arcuate (ring) scotoma
- Loss of peripheral vision especially of the superior and/or inferior hemifields
- Sparing of central vision
- Advanced stage
- Tunnel vision: further constriction of peripheral vision
- Total or near-total blindness: loss of peripheral and central vision with or without sparing of the temporal field
- Early-stage: arcuate or double arcuate (ring) scotoma
- Glaucomatous visual field defects: a characteristic pattern of visual field defects as a result of glaucomatous optic neuropathy
Treatment of acute angle-closure glaucoma
Acute angle-closure glaucoma is an emergency and should be initially managed with IOP-decreasing medications that have a rapid onset of action. Once IOP has decreased, patients should undergo a definitive procedure as soon as possible to prevent recurrence.
- Emergency ophthalmology consultation
- Place the patient in a supine position.
- Ensure the contralateral eye has been evaluated for urgent treatment, even if it is asymptomatic.
- Administer supportive care as needed.
- Analgesics (see pain management)
- Antiemetics (e.g., ondansetron )
Initial pharmacotherapy for glaucoma
- Indication: initiate in all patients as soon as a diagnosis of acute angle-closure glaucoma is made.
- Initial pharmacological regimen: There is currently no standardized recommendation for empiric management of acute angle-closure glaucoma. The following regimen may be followed with due consideration of any comorbidities.
- Topical ophthalmic therapy. Administer the following eye drops in succession, one minute apart:
- Direct parasympathomimetic: pilocarpine
- Alpha-2 agonist: apraclonidine
- Beta blocker: timolol
- PLUS a systemic carbonic anhydrase inhibitor
- Acetazolamide
- OR methazolamide
- Consider also the following alternatives:
- Betaxolol
- Brinzolamide
- Dorzolamide
- Levobunolol
- Brimonidine
- Topical ophthalmic therapy. Administer the following eye drops in succession, one minute apart:
- If IOP is still elevated after 30–60 minutes: The following should be given only under the guidance of an ophthalmologist.
- Repeat eye drops from above up to three times.
- Consider a systemic hyperosmotic agent if IOP remains high after 60 minutes of initiating therapy.
- In patients with nausea: IV mannitol
- In patients without significant nausea
- Nondiabetic patients: oral glycerine
- Diabetic patients: oral isosorbide
- If IOP is decreasing: Examine for other signs of resolution of the acute attack.
- Symptomatic improvement (decreased pain and nausea; improvement of vision)
- Clear cornea (resolution of corneal edema)
- Decreased conjunctival hyperemia
- Normal pupillary size and reaction

Urgent interventional therapy
- Anterior chamber paracentesis
- Indication: Consider for vision-threatening elevation in IOP refractory to medical management.
- Procedure: controlled drainage of some of the aqueous humor through an opening created at the limbus
- Important consideration: Anterior chamber paracentesis is a temporizing measure; patients will still require definitive treatment with laser peripheral iridotomy.
- Urgent laser peripheral iridotomy
- Indication: all patients within 24–48 hours of resolution of the acute attack
Topical pilocarpine becomes effective only once IOP decreases to < 40 mm Hg.
Acute management checklist for acute angle-closure glaucoma
- Emergency ophthalmology consult
- Place patient in a supine position.
- Initiate pharmacotherapy as soon as the diagnosis is made.
- Consider hyperosmotic pharmacotherapy if initial treatment is unsuccessful.
- Supportive care, as needed
- Admit patient or transfer to a hospital with ophthalmology department capable of performing LPI.
Treatment of chronic primary angle-closure glaucoma
Chronic angle-closure glaucoma with pupillary block should be initially managed with laser surgery (e.g., peripheral iridotomy) or open surgery (iridectomy) to prevent the progression of glaucomatous optic neuropathy
and consequent visual field loss. Long-term pharmacotherapy is required if IOP elevation is refractory to the intervention or in patients without pupillary block.
- Laser peripheral iridotomy (LPI): preferred first-line therapy if pupillary block is suspected
- Maintenance pharmacotherapy: similar to that of open-angle glaucoma
- Indications
- Persistently elevated IOP despite iridotomy
- Chronic primary angle-closure glaucoma without pupillary block
- Indications
Interventional therapy
Acute angle-closure glaucoma and chronic primary angle-closure with pupillary block
- Laser peripheral iridotomy (LPI)
- Indications
- Standard of care for acute angle-closure glaucoma as soon as the acute attack is resolved and the cornea becomes clear
- First-line therapy for chronic primary angle-closure glaucoma with pupillary block
- Prophylactic therapy for the contralateral eye in patients with unilateral angle-closure glaucoma
- Procedure: the creation of a hole in the peripheral iris to allow aqueous humor to bypass the pupillary block using a laser (preferably neodymium:YAG)
- Disadvantages
- Risk of closure of the iridotomy
- Post-operative IOP
- Laser burn
- Transient blurred vision
- Progression to cataract formation
- Indications
- Laser peripheral iridoplasty (gonioplasty)
- Indication: persistently elevated IOP despite a patent LPI
- Procedure: creation of burn contractures in the peripheral iris with an argon laser to pull the peripheral iris away from the iridocorneal angle
- Surgical peripheral iridectomy
- Indication: an alternative to LPI in patients with acute/chronic angle-closure glaucoma with pupillary block
- Procedure: the surgical excision of a small amount of iris tissue to allow for aqueous flow
- Disadvantages
- Costly
- Postoperative recovery period
- Surgical complications E.g., infection, wound leak, zonular or lens disruption
Chronic primary angle-closure glaucoma without pupillary block
Secondary angle-closure glaucoma
- Surgery for the underlying cause, such as:
- Cataract surgery
- Goniosynechialysis
Childhood Glaucoma
Definition
Any abnormal increase in intraocular pressure during the first years of life will cause dilatation of the wall of the globe, and especially of the cornea. The result is a characteristic, abnormally large eye (buphthalmos) with a progressive increase in corneal diameter. This is also referred to as hydrophthalmos or hydrophthalmia.
Epidemiology
Glaucomas in children occur once every 12000–18000 births and account for about 1% of all glaucomas. Primary congenital glaucoma is an inherited autosomal recessive disorder. It is bilateral in approximately 70% of all cases; boys are affected in approximately 70% of all cases; and glaucoma manifests itself before the age of six months in approximately 70% of all cases. Today there is widespread public awareness of glaucoma in adults. Unfortunately, this does not yet apply to glaucoma in children.
Symptoms
Classic signs include photophobia, epiphora, corneal opacification, and unilateral or bilateral enlargement of the cornea. These changes may be present from birth (in congenital glaucoma) or may develop shortly after birth or during the first few years of life. Children with this disorder are irritable, poor eaters, and rub their eyes often. The behavior of some children may lead one to suspect mental retardation. Physicians should be alert to parents who boast bout their child’s “big beautiful eyes” and should measure intraocular pressure. It is essential to diagnose the disorder as early in the child’s life as possible to minimize the risk of loss of or irreparable damage to the child’s vision.
Diagnostic considerations
These examinations may be performed without general anesthesia in many children. However, general anesthesia will occasionally be necessary to confirm the diagnosis especially in older children
Prognosis
Goniotomies and trabeculotomies are not always successful. Even after apparently successful initial trabecular surgery, these children require a lifetime of follow-up examinations (initially several times a year and later once every year) as elevated intraocular pressure can recur, in which case repeat goniotomy or trabeculotomy is indicated.
 تخطي إلى المحتوى
تخطي إلى المحتوى
