written by Prof. Khalil alsalem as a comentary on Dr. Alaa khamees lecture.
Optical coherence tomography (OCT) is, for most of us, the default structural test when the optic nerve is in question—whether we’re tracking glaucoma, differentiating papilledema from pseudopapilledema, or following optic neuritis. Yet the apparent simplicity of a colored TSNIT curve can hide a surprising number of assumptions and pitfalls. This article walks from the physics-to-picture basics through scan protocols, metrics, artifacts, and finally, pattern recognition across the major optic neuropathies. The goal is to make you unreasonably confident about what the machine is showing—and what it isn’t.
From Physics to Picture: What Exactly Are We Looking At? how Optic nerve OCT is Built?
Spectral-domain (SD) and swept-source (SS) OCT both measure echo time delay and intensity of back-scattered low-coherence light. A single axial reflectivity profile (A-scan) is a column of depth information. Lining A-scans side-by-side generates a cross-section (B-scan). Stacking B-scans yields a 3D volume. SS-OCT (longer central wavelength, ~1050 nm) penetrates deeper with less sensitivity roll-off, which is helpful at the lamina cribrosa and in highly myopic eyes. SD-OCT (840–870 nm) remains the clinical workhorse, with abundant longitudinal data and robust glaucoma software.
Layers that matter around the disc (Macular OCT , related to optic disc disease)
RNFL (retinal nerve fiber layer): unmyelinated ganglion cell axons; the classic peripapillary metric in glaucoma.
GCL/IPL (ganglion cell + inner plexiform layers): tracked as GCIPL/GCC in macular scans; often shows earlier change than RNFL in glaucoma and some non-glaucomatous neuropathies.
Bruch’s membrane opening (BMO) and minimum rim width (BMO-MRW): anatomically-anchored neuroretinal rim metric measured from the BMO to the internal limiting membrane along the shortest line—less confounded by disc size/tilt than “clinical rim.”
Lamina cribrosa (LC): visible with enhanced depth imaging (EDI) or SS-OCT in many eyes; research-heavy but increasingly clinically relevant in atypical cupping.
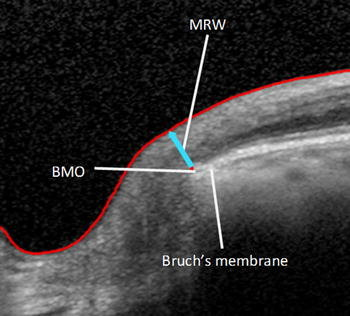
Key takeaways.
Peripapillary RNFL thickness reflects axonal content; 2) Macular GCIPL/GCC reflects somas + dendrites; 3) BMO-MRW reflects true rim tissue independent of subjective disc margins. You should interpret all three in concert.
Scan Protocols and What Each Is Good For in optic nerve OCT
Peripapillary RNFL circle scan (the “3.4-mm circle”).
A ring scan around the disc at an instrument-defined radius (nominally 3.4 mm) produces the familiar TSNIT curve (Temporal–Superior–Nasal–Inferior–Temporal). Device software compares thicknesses in 12 clock hours, quadrants, and global average to a normative database. Fast, repeatable, progression-friendly. Vulnerable to magnification error in high myopia and to circle misplacement if the optic disc center is mis-registered.
Macular cube (GCIPL/GCC analysis).
A dense volumetric scan over ~6×6 mm centered on the fovea. Provides average and sectoral ganglion-cell complex thickness. Particularly helpful when RNFL is near the “floor” (advanced glaucoma), in small crowded discs, or when peripapillary anatomy is distorted (tilt, peripapillary atrophy).
BMO-MRW radial scans.
A stack of radial B-scans centered on the ONH used to segment BMO and compute minimum rim width. Better accommodates disc size extremes, tilt, and torsion. If you’re dealing with very large or very small discs—or trying to adjudicate “large cup vs real loss”—BMO-MRW is your friend.
Enhanced Depth Imaging (EDI) / Swept-Source for LC and drusen.
EDI on SD-OCT or SS-OCT improves visualization of the LC and deep ONH and helps detect optic disc drusen (ODD) and peripapillary hyperreflective ovoid mass-like structures (PHOMS). Helpful in pseudopapilledema workups and atypical cupping.
OCT Angiography (OCTA) around the disc.
Peripapillary capillary density and flux indices (e.g., radial peripapillary capillaries) are often reduced in glaucoma and ischemic optic neuropathies. OCTA is sensitive but currently adjunctive; interpret alongside structure and function because it is influenced by motion, projection, and systemic hemodynamics.
Normative Databases in optic nerve OCT: Useful—but Know Their Blind Spots
Normative color maps (green/yellow/red) are calculated from manufacturer-specific reference populations. Limitations you must remember:
Axial length and refractive error: High myopia skews RNFL thinner and alters topography; many databases under-represent −6 D and beyond.
Disc size and tilt: Larger discs can look “abnormally thin” by RNFL while actually being normal; smaller discs the reverse. BMO-MRW reduces this bias.
Ethnicity/age: Databases vary, and age-adjustment is imperfect at the extremes.
Disease not in database: Normative maps do not “recognize disease”—they highlight deviation from an average. Use the raw B-scans and en face views to adjudicate.
Bottom line: color is a clue, not a verdict. Always verify on the B-scan and check the sectoral pattern for biological plausibility.
Artifacts of optic nerve OCT and How to Catch Them in Seconds
Signal strength / media clarity.
Poor tear film, cataract, or small pupils reduce signal, causing diffuse RNFL thinning and segmentation errors. Redo after lubricating, blinking, or improving fixation. Most devices flag low signal strength—treat global red maps with suspicion when signal is borderline.
Segmentation errors.
Look for the segmentation lines on the B-scan. Mislabeling ILM or RNFL boundaries can add or subtract tens of microns. Common in epiretinal membranes, peripapillary atrophy, drusen, and high myopia.
Circle mis-centering and magnification.
If the ring is off-center or its physical radius differs because of axial length, sectoral RNFLs shift artificially. On some platforms you can enable axial-length-based scaling (Bennett/Littmann correction); on others, rely more on macular GCIPL and BMO-MRW in high myopia.
Motion and blink lines.
Horizontal discontinuities in the TSNIT or macular maps suggest motion. Repeat the scan; ask the patient to breathe normally and keep lids relaxed.
Floor effect.
In advanced glaucoma, RNFL asymptotes to ~40–50 µm (device-dependent). Progression then hides in macular GCIPL or rim metrics; a “stable” RNFL in a very damaged eye doesn’t mean stability.
Peripapillary atrophy (PPA).
Segmentation lines can wander into PPA, producing spurious thin sectors (often temporal). Inspect the B-scan near the beta-PPA edge and consider BMO-MRW.
A good daily habit: always (1) check signal, (2) review B-scans with segmentation overlays, (3) verify ring centering, (4) reconcile with disc size and axial length, (5) cross-check RNFL vs GCIPL/BMO-MRW.
Metrics You Should Actually Use
Global RNFL average: Easy to track; robust longitudinally. Beware floor effect and myopia.
Quadrants and clock hours: Inferior and superior sectors are most glaucoma-sensitive; temporal sectors matter in optic neuritis and chiasmal lesions.
TSNIT profile: The shape matters as much as absolute thickness; localized notches in the superotemporal/inferotemporal peaks are classic for early glaucoma.
Macular GCIPL/GCC: Strong for early detection and advanced follow-up; pay attention to hemifield asymmetry.
BMO-MRW: Less dependent on disc size/tilt; complements RNFL in ambiguous discs.
Structure-function mapping: Superotemporal RNFL ↔ inferior arcuate field; macular GCIPL maps to central 10–24° field. Hood’s structure-function framework is useful: localized macular GCIPL defects often predict paracentral field loss.
Progression analysis:
Event-based: Compare to baseline (eye turns “red” beyond test–retest variability).
Trend-based: Linear regression over time (µm/year).
Use both: event analysis flags sudden change; trend analysis quantifies rate. Ensure comparable signal quality and the same device across visits.
Inter-Device Differences in optic nerve OCT (and Why You Shouldn’t Mix)
Each platform (e.g., Zeiss, Heidelberg, Topcon, Nidek, Optovue) uses different scan geometries, segmentation rules, and normative data. RNFL values are not interchangeable across devices. Stick to one platform for progression. If you must switch (e.g., clinic upgrade), establish a new baseline on the new device and rely more on qualitative patterns, GCIPL, and VF trends during the bridging period.
Special Populations: Myopia, Tilted Discs, Large/Small Discs
High myopia (axial length ≥ 26 mm)
Thinner global RNFL, temporal shift in peaks, wide PPA, frequent segmentation errors.
Rely more on macular GCIPL and BMO-MRW. Consider enabling axial-length scaling if available.
Be cautious with red/green maps; “abnormal” may be normal for the eye.
Tilted discs / torsion
Sectoral RNFL may appear pathologic because the circle intercepts different fiber trajectories.
BMO-MRW helps; inspect en face RNFL probability maps for arcuate-shaped defects rather than color alone.
Giant discs / small discs
In very large discs, RNFL can look thin and color maps go yellow/red without true disease.
BMO-MRW is less biased; always reconcile with clinical rim and vessel kinking.
OCT A Around the Optic Nerve: What It Adds
Peripapillary vessel density (VD) and perfusion metrics decline in glaucoma, often mirroring structural loss. In non-arteritic ischemic optic neuropathy (NAION), peripapillary capillary dropout corresponds to RNFL/GCIPL loss and can help distinguish NAION from optic neuritis (which may have relatively preserved peripapillary flow in the acute phase). In papilledema, superficial peripapillary capillaries may appear congested rather than dropped out. Limitations: motion artifacts, projection, segmentation on swollen nerves, and systemic vasodynamics. Treat OCTA as supportive evidence, not a sole diagnostic.
Disease Patterns on optic nerve OCT : A Practical Atlas
A. Glaucoma
Early disease
RNFL: Focal thinning in superotemporal or inferotemporal sectors; localized notches on TSNIT peaks.
GCIPL: Hemifield or wedge-shaped defects corresponding to arcuate regions; may precede peripapillary RNFL change in some eyes.
BMO-MRW: Focal rim thinning aligned with RNFL defect.
OCTA: Peripapillary capillary density drop overlapping the structural defect.
Moderate disease
RNFL: Sectoral thinning coalesces; average RNFL declines at a rate often between ~0.5–2 µm/year (highly variable).
GCIPL: Clear hemifield loss with parafoveal involvement; structure–function agreement improves.
BMO-MRW: Rim loss corroborates peripapillary thinning.
Advanced disease
RNFL: Approaches floor (~40–50 µm).
GCIPL: Better dynamic range for monitoring; look for continued parafoveal thinning tied to central field loss.
Strategy: Switch progression emphasis to GCIPL, BMO-MRW, and 10-2 VF testing; review for “split fixation.”
Red flags for “not typical glaucoma”
Predominant temporal RNFL/GCIPL loss with central/cecocentral scotoma (consider toxic/nutritional/hereditary).
Marked vertical midline macular pattern (think chiasmal).
Diffuse RNFL loss without cupping or with pallor > cupping (compressive, inflammatory).
B. Optic Neuritis (demyelinating)
Acute (first weeks): RNFL can be swollen (pseudo-thick). GCIPL may still be normal. OCTA usually not diagnostic.
3–6 months:
GCIPL: Thins early (retrograde degeneration), often detectable by 6–8 weeks.
RNFL: Thins subsequently (by ~3–6 months).
The temporal sector and papillomacular bundle are often most affected; asymmetric GCIPL thinning with relatively preserved rim/cup distinguishes from glaucoma. Use GCIPL to document permanent axonal loss and to prognosticate visual recovery alongside VF and VEP.
C. NAION
Acute: Disc edema; RNFL elevated globally or sectorally; segmentation unreliable.
Chronic (months):
- RNFL: Sectoral loss usually inferior or superior corresponding to altitudinal VF; often more abrupt borders than glaucoma.
- GCIPL: Sectoral loss with relative sparing of the macular area not involved in the altitudinal defect.
- OCTA: Peripapillary capillary dropout in affected sectors.
Clues vs glaucoma: smaller or absent cupping, crowded fellow disc, altitudinal pattern, and history of acute event.
D. Compressive Optic Neuropathy (chiasmal or retrobulbar)
Macular GCIPL: Bitemporal hemiretinal thinning in chiasmal lesions (nasal macular fibers cross at chiasm → temporal macular loss). The vertical midline split in en face/GCIPL maps is highly suggestive.
RNFL: Nasal/temporal sector changes depending on lesion site; often discordant with “glaucoma pattern.”
Use the OCT pattern to motivate neuroimaging when the clinical picture is equivocal.
E. Toxic/Nutritional and Hereditary Optic Neuropathies
Pattern: Predominant temporal RNFL and central/temporal GCIPL thinning → cecocentral scotoma clinically (e.g., ethambutol toxicity, Leber hereditary optic neuropathy after the acute swelling phase). OCT tracks progression and recovery (if any) after cessation of the agent.
F. Papilledema vs Pseudopapilledema (ODD, crowded discs)
Papilledema (true swelling)
RNFL: Diffuse elevation; peripapillary total retinal thickness (TRT) elevated.
Sub-retinal hyporeflective space (peripapillary) on B-scan common.
OCTA: Superficial peripapillary capillary congestion rather than dropout early.
Longitudinal: RNFL should fall with treatment; watch for secondary GCIPL thinning indicating axonal loss.
Optic disc drusen (ODD) / pseudopapilledema
EDI/SS-OCT: Hyperreflective ovoid deposits in the prelaminar/within ONH; may be superficial or buried.
PHOMS: Peripapillary hyperreflective ovoid mass-like structures can occur in ODD and other crowded discs; not pathognomonic.
RNFL: May be thick (“crowded”) early but can thin over time from chronic axon loss; sectoral defects are possible.
When uncertain, combine EDI-OCT with B-scan ultrasound and autofluorescence; serial OCT helps distinguish dynamic swelling (papilledema) from static elevation (ODD).
G. Traumatic Optic Neuropathy
Initial OCT may be normal. By 4–8 weeks, GCIPL thinning emerges; RNFL follows. OCT helps counsel on prognosis and objectify axonal loss.
Putting It Together in Clinic: A Stepwise Interpretation Workflow
Is the scan usable?
Signal strength adequate? Motion/blink lines absent? Ring well centered?
Segmentation lines accurate on representative B-scans?
What is the disc phenotype?
Size, tilt, torsion, PPA, axial length. Decide a priori how much you’ll “trust” RNFL vs GCIPL vs BMO-MRW.
Synthesize the three pillars
RNFL (where is thinning? shape of TSNIT?), GCIPL (hemifield/macular maps), BMO-MRW (rim integrity).
Check agreement across metrics and with the optic disc exam and VF.
Consider disease-specific patterns
Glaucoma arcuates? Temporal/cecocentral (toxic/hereditary)? Vertical split (chiasmal)? Altitudinal (NAION)? Diffuse with disc swelling (papilledema)?
Plan follow-up and progression strategy
Choose the same device, same protocol, optimize signal, and set baselines for RNFL, GCIPL, and (if available) BMO-MRW.
Use event + trend analysis; switch emphasis to GCIPL/BMO-MRW when RNFL nears floor.
Reporting Tips (What to Put in Your Note)
Acquisition quality: “Signal 8/10, well-centered ring, no motion; segmentation verified.”
Key metrics: Global/sectoral RNFL, macular GCIPL average + hemifield asymmetry, BMO-MRW global/sectoral if available.
Pattern statement: “Inferotemporal RNFL notch with concordant inferonasal macular GCIPL loss → matches superior arcuate VF defect.”
Comparative trend: “Since 2023 baseline, RNFL −1.1 µm/yr; GCIPL −0.6 µm/yr (both significant; p<0.05).”
Caveats: “High myopia (AL 27.8 mm); interpret color maps cautiously; progression adjudicated primarily with macular GCIPL/BMO-MRW.”
Practical Pearls You’ll Use Tomorrow
If the VF is suspicious but RNFL is “green,” check GCIPL. Early parafoveal loss often lives in the macula first.
In high myopia, normalize by using macular GCIPL and BMO-MRW. Turn on axial-length corrections when available.
“Stable” RNFL in advanced glaucoma may be a floor illusion. Follow GCIPL and central 10-2.
OCTA is additive, not decisive. Use it to support, not to replace, structure + function.
When disc looks big and RNFL looks “thin,” ask BMO-MRW. It often rescues you from over-calling glaucoma in large discs.
Suspect compressive disease with a vertical macular split. GCIPL hemifield maps can trigger the MRI that saves vision.
Distinguish papilledema from ODD with EDI-OCT and time. Drusen sit still; papilledema evolves.
Common Pitfalls in optic nerve OCT (and Quick Fixes)
Pitfall: Believing the color map.
Fix: Read the B-scan; confirm segmentation; cross-check with macular and rim metrics.
Pitfall: Mixing devices for progression.
Fix: Don’t. If forced, establish a new baseline.
Pitfall: Ignoring axial length.
Fix: Document AL or refractive error; preferentially weigh GCIPL/BMO-MRW in high myopia.
Pitfall: Calling ODD “papilledema” on one scan.
Fix: EDI-OCT/US, autofluorescence, and serial change; look for PHOMS but remember it’s not specific.
Pitfall: Overreading diffuse RNFL loss with poor signal.
Fix: Reacquire after improving surface optics (blink, lubricate, adjust chin rest).
Where the is optic nerve OCT Going
Standardization around BMO-MRW is increasing because it is anatomy-anchored and less biased by disc size/tilt.
Deep ONH imaging with SS-OCT may improve lamina-based biomarkers (beam displacement, pore morphology).
Machine learning is moving from classification to explainable sectoral risk and rate predictions across multimodal inputs (RNFL + GCIPL + VF + OCTA).
Axial-length aware analytics will better handle the global wave of myopia.
For now, the most impactful upgrades you can make are workflow: always verify segmentation, triangulate RNFL with GCIPL and BMO-MRW, and interpret within the clinical phenotype.
 تخطي إلى المحتوى
تخطي إلى المحتوى


الله يعطيكم العافية
محاضرة قيمة جدا